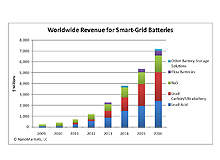
Utility-scale battery use is looking at an almost exponential increase over the next five years as more intermittent renewable power sources like wind and solar come on line. When renewables supply above ten to 15% of grid power, they need backup storage or the grid itself can become unstable.
Batteries have much lower running costs – and environmental impacts – than natural gas and diesel-fuelled reserves. With pumped hydro and compressed air energy storage (CAES) only possible in a few suitable geographic regions, there is still no real general alternative to batteries for storing tens of MW.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
They will particularly be needed as smart grids start to incorporate renewable supplies.
Deep-cycle batteries are already universal in off-grid residential and industrial renewables systems.
Even industrial systems are still only relatively small scale, though, for powering traffic signals, video security, telecoms, radio communications and pipeline monitoring / control. With a serious upscaling for utility use, banks of batteries can now store power generated during low demand and pass it on up to eight hours later during high demand. This can give utilities better than five-year repayment times by reducing peak power construction, transmission and distribution costs.
What little grid storage there has been so far has mainly come from lead-acid batteries, but improvements in deep-cycling performance will be needed for large-scale renewable storage.
There has therefore been increasing interest in sodium sulphur (NaS) batteries and, more recently still, flow batteries.
These will compete even more strongly as prices start to drop from volume manufacturing. Lead-carbon looks the technology to watch, promising to solve the major problems of lead-acid batteries.
Lead-acid has so far dominated
The familiar ‘flooded’ lead acid battery has porous lead (Pb) at the negative electrode, lead dioxide (PbO2) at the positive electrode, and dilute sulphuric acid (H2SO4) as the electrolyte in between. Connecting a load to the terminals produces lead sulphate at the negative terminal and lead sulphate and water at the positive terminal, with electrons flowing through the load.
Recharging the battery reverses the process, although most lead-acid batteries can survive less than 1,000 complete charge / discharge cycles unless they are derated / oversized. When a flooded lead-acid battery is nearly fully charged, the charging current starts to electrolyse the water and produce hydrogen and oxygen. Besides producing volatile hydrogen gases, this means the battery must be maintained by adding water.
Valve regulated lead acid (VRLA) types solve these last two problems – they are sealed and need no special ventilation or watering. GNB Industrial Power (a division of Exide Technologies) is one of the largest lead-acid battery manufacturers, with a broad range of high-cycling batteries for photovoltaic systems.
GNB’s Director of Utility Accounts Sean Blaylock remarks that the main issues for lead-acid batteries are temperature and charging behaviour. Going above 25°C reduces lifetimes, while overcharging increases positive grid corrosion (life reduction), and undercharging increases plate sulphation (capacity reduction).
Maintenance mainly involves monitoring the battery bank to maximise life and performance, and replacing batteries and cells that are outside performance specification.
Worker safety has to be paramount in an environment with high voltages and hydrogen evolution. Operators need to gather quarterly or half-yearly temperature, voltage, and ohmic /conductance readings.
Large battery systems are already successfully backing up utility power systems in remote locations. A one MW turnkey battery energy storage system (BESS) designed by a GNB / General Electric consortium for Metlakatla Power & Light Utility (Alaska) uses VRLA batteries to back up hydroelectric plant.
The 1.4 MWh system provides power system stabilisation, frequency control and VAR support. The battery room holds 1,134 cells which have replaced a three MW diesel generator. Costing $1.5 million in 1997, the system is still working. It paid for itself within three years and saved Metlakatla more than $6.5 million in 12 years of operation, along with nearly 59,000 tons of CO2 emissions.
As large scale renewable storage applications grow, the demand for improved deep cycling performance will be needed. As a result, NaS batteries are finding a place in the market.
NaS and now flow batteries take market share
NaS batteries withstand more discharging cycles (around 2,500 at 100% depth of discharge). They have higher capital but lower balance-of-plant costs (including power electronics).
They also maintain their good charge / discharge efficiencies at high state of charge (SOC) – although the charge / discharge efficiency of lead acid batteries can reach 90% overall, it can be low (just more than 50%) at high SOC.
NaS was originally developed by NGK Insulators and Tokyo Electrical Power Company, and the batteries are widely used for grid storage in Japan.
They have liquid (molten) sodium at the negative electrode, sulphur at the positive electrode and a BASE (beta-alumina solid electrolyte) ceramic separating the two. When a load is connected to the terminals, sodium ions pass through the solid electrolyte to form sodium polysulphide at the positive sulphur electrode – again causing electrons to flow in the load.
The technology holds the record for the largest working battery system – NGK’s 34 MW battery at Rokkasho, Japan. This supplies 220 MWh, regulating and providing nearly seven hours back-up for a 51 MW wind farm.
Prices should drop as volume manufacture increases – General Electric has announced it will be producing sodium-based batteries in its Niskayuna factory in New York, for example.
One drawback is the operating temperature of 300°C. The heat produced by charging and discharging cycles is enough to maintain temperatures in the large battery banks, though, and they need no external heat source. Lower-temperature versions – and different types of liquid metal battery – are in development.
More recent still are flow batteries, where the electrolyte is stored externally and pumped through the cells of the reactor. The most widely commercialised type so far has been the vanadium redox (reduction-oxidation) flow battery, which has been used in several countries, particularly Japan.
This has two electrolytes – both vanadium based but using vanadium ions that have different charges – separated by a proton exchange membrane. Capacity can be scaled up simply by increasing the size of the storage tanks, and development models have already hit 10,000 charges / discharges.
Sumitomo Electric Industries has several vanadium RFB installations operating, including a four MW wind-farm-based plant at Sapporo. Other possible materials include zinc / bromine, cerium / zinc and iron / chrome.
Li-ion batteries on the fringe
Grid-scale lithium-ion batteries have so far not really competed because of their higher cost, although prices are dropping as production volumes rise. A123 Systems has a Li-ion battery system using its proprietary Nanophosphate lithium-ion chemistry. The systems are supplied as modules, each with two MW power and 500 kWh energy capacity. They can scale up to above 200 MW, and withstand from 10,000 to more than 100,000 cycles depending on the throughput.
By the end of 2011, A123 Systems expects to have more than 100MW installed worldwide. It has with AES Gener for example installed 12 MW spinning reserve at the Los Andes substation in the Atacama Desert in Chile. The system continuously monitors the power condition and almost instantaneously provides up to 12 MW for 15 minutes if there is a significant frequency deviation.
As well as improving operational efficiency and reducing emissions, the system has increased AES Gener’s revenue because the system replaces unpaid reserve.
A similar 20 MW spinning reserve project will back up a 500 MW power plant to be built in Northern Chile. Other projects include wind farms, and a 20 MW energy storage system for frequency regulation at the AES Westover coal-fired power station near Johnson City, N.Y.
With millions of hybrid and electric cars planned for our roads, these would seem an ideal solution to add distributed battery backup to grids. There have been proposals to charge them at night and discharge them back into the grid during the day. The lithium-ion batteries that look set to dominate this market could form a major part of a car’s cost, though, and the repeated charging / discharging could unacceptably reduce the battery’s lifetime. Another proposal is to use old batteries when hybrid cars start being replaced, when the batteries should still have 70% of their capacity left. This will take at least a decade before it can make any appreciable difference, though.
By that time, lead-carbon batteries promise to dominate the market, quadrupling the energy density of lead-acid batteries and with much better lifetimes for much lower prices than NaS types. Axion Power has laboratory prototypes that withstand more than 2,000 complete charge / discharge cycles. They are similar to lead-acid types but instead simple sponge lead plates, the negative electrodes are five-layer assemblies having a carbon electrode, corrosion barrier, current collector, second corrosion barrier and second carbon electrode. The very high surface area of the activated carbon effectively gives a battery-supercapacitor hybrid.
The key to Axion’s success will lie in the leap to high-volume production. That is particularly true for the huge market promised by microgrids. Here, battery-based renewable energy could bring stable and reliable power to the above 80% of the world’s population who live in rural areas with no or unreliable access to electricity. In India, China and elsewhere, microgrids will reduce infrastructure costs and transmission losses. They will likely include different combinations of fuel cells, supercapacitors and batteries – with lead-carbon types as a major contender if they fulfil their early promise.





